2.5 Identify the chemical elements present in carbohydrates, proteins and lipids
(fats and oils).
|
Carbohydrates:
|
Proteins:
|
Lipids (fats and oils):
|
2.6 Describe the structure of carbohydrates, proteins and lipids as large molecules made up from smaller basic units.
|
Carbohydrates:
Simple sugars (monosaccharides) such as glucose are the basic carbohydrates. They join together to make more complex (disaccharides) and (polysaccharides). The bond's holding them together are called glycosidic bonds. Examples:
|
2.7 Describe the tests for glucose and starch.
There are two simple tests which can be conducted to find out if Glucose or Starch are present in a substance or solution.
|
Glucose
If Benedict's colour changes from Blue > Red = Positive If Benedict's colour does not change = Negative If the solution is positive, you can see the colour change from Green > Yellow > Red. Benedict's test can also be used to test how much Glucose is present. |
Starch
If Iodine colour changes from Yellow > Blue = Positive If Iodine colour does not change = Negative |
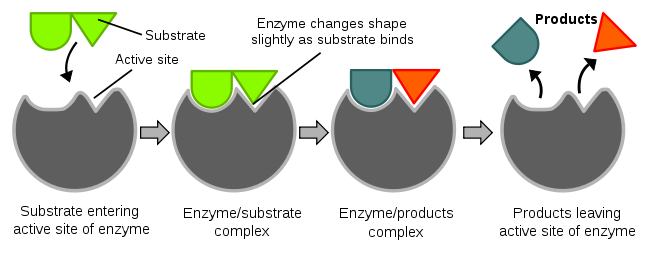
2.8 Understand the role of enzymes as biological catalysts in metabolic
reactions.
Just like in chemistry, a catalyst speeds up metabolic reactions.
This is because the enzymes lower the activation energy of a reaction, Making it faster.
They are unchanged from beginning to end of a reaction.
This is because the enzymes lower the activation energy of a reaction, Making it faster.
They are unchanged from beginning to end of a reaction.
2.9 Understand how the functioning of enzymes can be affected by changes in
temperature, including changes due to change in active site.
- As heat increases kinetic energy increases,
- This means enzymes and substrates move around more so they are more likely to collide and bind,
- This means that as heat increases so does the amount of reactions an enzyme catalyses,
- However if the temperatures are too high it will denature enzymes, so they can't function.
- This is because the energy breaks the bonds that hold the shape of the enzyme.
- Without these the structure will be distorted, which will mean the active site won't be able to bind with the substrate to break it down.
|
The active site is the area on an enzyme that binds with a substrate to break it down. (It is the lock in the lock and key theory).
Optimum temperature is in between these two scenarios. Where there is lots of energy from heat but not enough to denature the enzyme. It is different for different enzymes. |
2.11 Describe experiments to investigate how enzyme activity can be affected by changes in temperature.
- Put starch into a test tube. Either heat or cool it,
- Add amylase (the catalyst),
- Spot this mixture on a spotting tile, and add iodine,
- Time how long it takes for the iodine to stop being blue black
- Repeat at different temperatures and compare
When the iodine stops being blue/black there is no starch present, so it must have been digested by the enzymes.